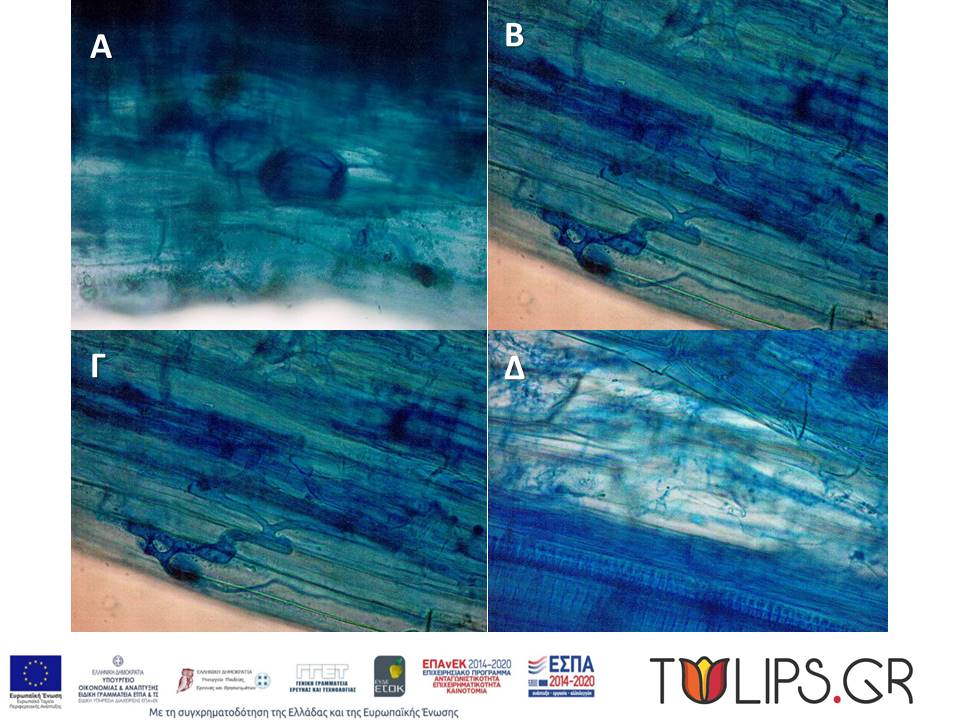
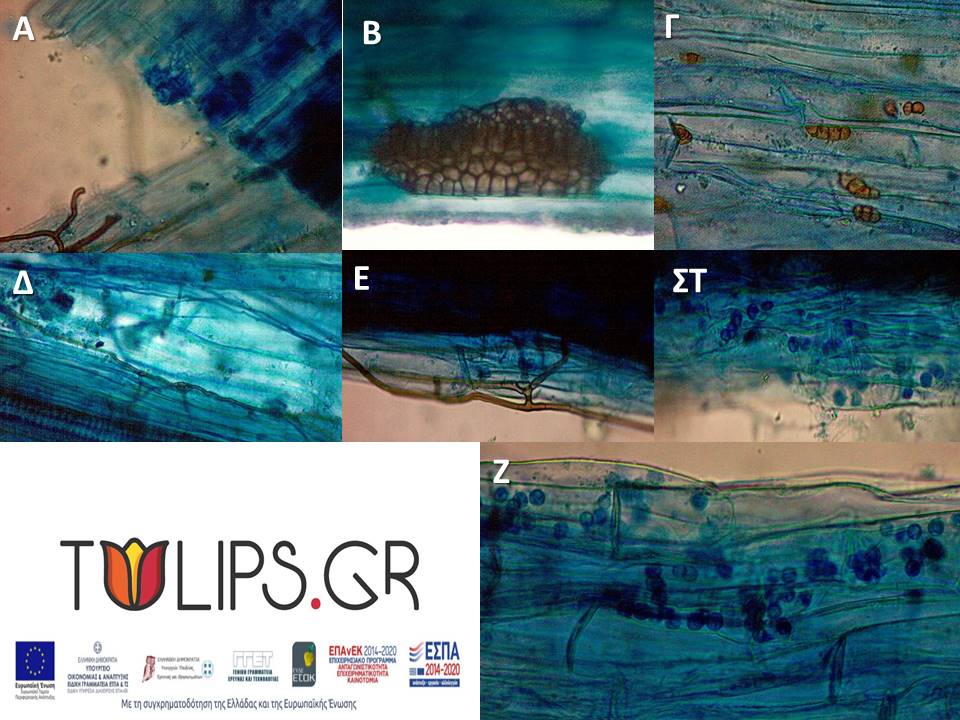
Root samples were placed in Petri dishes to remove soil residue with tweezers and on absorbent paper to evaporate the alcohol. Up to 0.1 g of up to 10 individuals of the same species per region were used for DNA extraction. In the case of small quantities, complex samples were created. The remaining volume was placed in centrifuge tubes for staining and counting mycorrhizal colonization under a microscope.
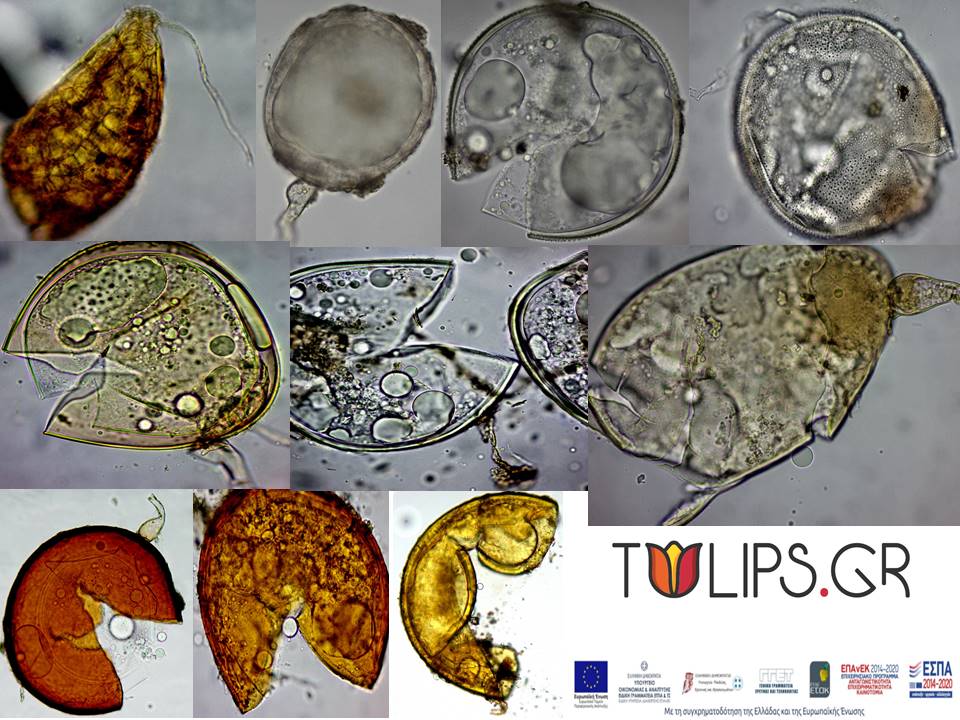
As the number of individuals in which colonization was counted varies according to the amount of available root after weighing/storage for DNA extraction, complex samples were created on a case-by-case basis. Spores were extracted from the rhizosphere for counting and homogeneous groups of spores per morphotype were kept in slides for microscopic observation (see representative photos below). Also, the rhizosphere soil was applied as a vaccine in potters with 2 kg of substrate (sterile mixture 1: 1 sand: vermiculite) with alfalfa, pentaneur and corn as plant-traps for vaccine propagation and spore isolation.